Instructor-Led Training Parameters
Course Highlights
- Instructor-led Online Training
- Project Based Learning
- Certified & Experienced Trainers
- Course Completion Certificate
- Lifetime e-Learning Access
- 24x7 After Training Support
Discover Veeva Clinical Operations Training Course Overview
Enroll in Veeva Clinical Operations training by Multisoft Systems to master Veeva Vault Clinical Suite, including CTMS, eTMF, and Study Startup. This comprehensive program equips professionals with practical knowledge, real-time use cases, and industry best practices to streamline clinical trial management and ensure regulatory compliance across global research environments.
Veeva Systems Clinical Operations Training by Multisoft Systems is designed to provide in-depth knowledge of the Veeva Vault Clinical Suite and its application in modern clinical trial management. The program focuses on core components such as Vault CTMS, Vault eTMF, Study Startup, and regulatory document management, enabling professionals to streamline end-to-end clinical operations processes effectively. This training covers system configuration, user roles, workflows, reporting, and compliance management aligned with global regulatory standards. Participants gain hands-on exposure to managing trial documentation, monitoring activities, site management, milestone tracking, and audit readiness within the Veeva environment. The course emphasizes industry best practices, process optimization, and digital transformation in clinical research.
Delivered by experienced industry trainers, the program includes real-world case studies and practical scenarios to help learners understand implementation challenges and solutions. It is ideal for clinical research professionals, clinical trial managers, regulatory associates, and life sciences IT specialists aiming to enhance operational efficiency and regulatory compliance using Veeva’s cloud-based platform.
Instructor-led Training Live Online Classes
Suitable batches for you
| May, 2026 | Weekdays | Mon-Fri | Enquire Now |
| Weekend | Sat-Sun | Enquire Now | |
| Jun, 2026 | Weekdays | Mon-Fri | Enquire Now |
| Weekend | Sat-Sun | Enquire Now |
Discover Veeva Clinical Operations Training Course curriculum
Curriculum Designed by Experts
Enroll in Veeva Clinical Operations training by Multisoft Systems to master Veeva Vault Clinical Suite, including CTMS, eTMF, and Study Startup. This comprehensive program equips professionals with practical knowledge, real-time use cases, and industry best practices to streamline clinical trial management and ensure regulatory compliance across global research environments.
Veeva Systems Clinical Operations Training by Multisoft Systems is designed to provide in-depth knowledge of the Veeva Vault Clinical Suite and its application in modern clinical trial management. The program focuses on core components such as Vault CTMS, Vault eTMF, Study Startup, and regulatory document management, enabling professionals to streamline end-to-end clinical operations processes effectively. This training covers system configuration, user roles, workflows, reporting, and compliance management aligned with global regulatory standards. Participants gain hands-on exposure to managing trial documentation, monitoring activities, site management, milestone tracking, and audit readiness within the Veeva environment. The course emphasizes industry best practices, process optimization, and digital transformation in clinical research.
Delivered by experienced industry trainers, the program includes real-world case studies and practical scenarios to help learners understand implementation challenges and solutions. It is ideal for clinical research professionals, clinical trial managers, regulatory associates, and life sciences IT specialists aiming to enhance operational efficiency and regulatory compliance using Veeva’s cloud-based platform.
- Understand the core functionality of Veeva Systems Vault Clinical Suite.
- Gain comprehensive knowledge of Vault CTMS, Vault eTMF, and Study Startup modules.
- Learn to configure workflows, user roles, and permissions within Veeva Vault.
- Develop skills to manage Trial Master File (TMF) documentation efficiently.
- Understand clinical trial lifecycle management from study startup to close-out.
- Track milestones, site performance, and monitoring activities using CTMS.
- Ensure regulatory compliance and maintain audit readiness standards.
- Generate reports and dashboards for effective clinical data tracking.
- Apply industry best practices for clinical operations process optimization.
- Enhance career opportunities in clinical research and life sciences IT.
Course Prerequisite
- Basic understanding of clinical research and clinical trial processes.
- Familiarity with clinical documentation and regulatory compliance concepts.
- Basic knowledge of IT systems and software navigation.
Course Target Audience
- Clinical Research Associates (CRAs)
- Clinical Trial Managers
- Regulatory Affairs Professionals
- Clinical Operations Specialists
- Life Sciences IT Professionals
- Project Managers in Clinical Research
- Quality Assurance and Compliance Officers
- Professionals aspiring to work in clinical trial management using Veeva Vault
Course Content
- Overview of Veeva Systems and Vault Clinical Suite
- Key concepts in clinical trials and operations
- Understanding the digital transformation in clinical research
 DOWNLOAD CURRICULUM
DOWNLOAD CURRICULUM
- Introduction to Vault platform and architecture
- Navigation, user interface, and role-based access
- Understanding dashboards and system features
 DOWNLOAD CURRICULUM
DOWNLOAD CURRICULUM
- Study setup, planning, and protocol management
- Overview of study milestones and monitoring activities
- Investigator and site tracking
 DOWNLOAD CURRICULUM
DOWNLOAD CURRICULUM
- Site management, enrollment tracking, and reporting
- Integration of CTMS with other Veeva modules
- Best practices for study execution
 DOWNLOAD CURRICULUM
DOWNLOAD CURRICULUM
- TMF structure and regulatory requirements
- Document types, indexing, and metadata
- Uploading and version control
 DOWNLOAD CURRICULUM
DOWNLOAD CURRICULUM
- Document review, approval, and workflow management
- Audit readiness and regulatory compliance
- Maintaining data integrity and quality standards
 DOWNLOAD CURRICULUM
DOWNLOAD CURRICULUM
- Site initiation, contracts, and budgets
- Regulatory submissions and approvals
- Workflow automation in Study Startup
 DOWNLOAD CURRICULUM
DOWNLOAD CURRICULUM
- Generating reports for CTMS and eTMF
- KPI tracking and dashboard customization
- Data analysis for operational decisions
 DOWNLOAD CURRICULUM
DOWNLOAD CURRICULUM
- Integrating CTMS, eTMF, and Study Startup modules
- Managing cross-functional workflows
- Collaboration and communication features
 DOWNLOAD CURRICULUM
DOWNLOAD CURRICULUM
- Hands-on exercises on all Veeva modules
- Real-world scenarios for problem-solving
- Applying best practices in clinical operations
 DOWNLOAD CURRICULUM
DOWNLOAD CURRICULUM
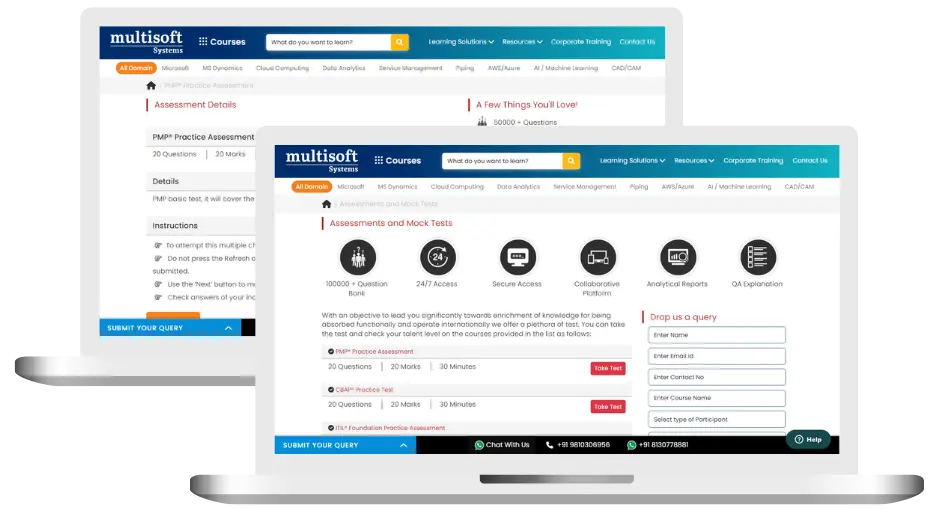
Discover Veeva Clinical Operations Training (MCQ) Assessment
This assessment tests understanding of course content through MCQ and short answers, analytical thinking, problem-solving abilities, and effective communication of ideas. Some Multisoft Assessment Features :
- User-friendly interface for easy navigation
- Secure login and authentication measures to protect data
- Automated scoring and grading to save time
- Time limits and countdown timers to manage duration.
Discover Veeva Clinical Operations Corporate Training
Employee training and development programs are essential to the success of businesses worldwide. With our best-in-class corporate trainings you can enhance employee productivity and increase efficiency of your organization. Created by global subject matter experts, we offer highest quality content that are tailored to match your company’s learning goals and budget.
Global Clients




Customized Training
Be it schedule, duration or course material, you can entirely customize the trainings depending on the learning requirements
Expert
Mentors
Be it schedule, duration or course material, you can entirely customize the trainings depending on the learning requirements
360º Learning Solution
Be it schedule, duration or course material, you can entirely customize the trainings depending on the learning requirements
Learning Assessment
Be it schedule, duration or course material, you can entirely customize the trainings depending on the learning requirements
Certification Training Achievements: Recognizing Professional Expertise
Multisoft Systems is the “one-top learning platform” for everyone. Get trained with certified industry experts and receive a globally-recognized training certificate. Some Multisoft Training Certificate Features :
- Globally recognized certificate
- Course ID & Course Name
- Certificate with Date of Issuance
- Name and Digital Signature of the Awardee
Discover Veeva Clinical Operations Training Trainer Profile
19+ Years Experienced
Our Discover Veeva Clinical Operations Training Corporate & Certification Program trainers bring 13+ years of proven industry expertise, delivering practical insights aligned with real project environments.
Trained 3950+ Professionals
Our expert trainers have successfully trained 3350+ professionals through structured, real-time training programs designed for industry readiness and career growth.
Certified Experts & Real-Time Project Learning
Build strong practical skills through live project-based training sessions led by certified industry experts with real-world experience.
Hands-on Learning Approach
Gain practical exposure through real-time scenarios, industry case studies, and hands-on assignments that simulate actual project challenges.
Certification Training Guidance
Receive expert support to prepare effectively, practice strategically, and confidently achieve globally recognized certification success.
Customized Training Delivery
Flexible training approach tailored to individual learning goals, skill levels, and evolving industry requirements for maximum effectiveness.
Discover Veeva Clinical Operations Training FAQ's
It is a comprehensive program that teaches professionals how to manage clinical trials using Veeva Systems Vault Clinical Suite, including CTMS, eTMF, and Study Startup modules.
What Attendees are Saying
Our clients love working with us! They appreciate our expertise, excellent communication, and exceptional results. Trustworthy partners for business success.
Share Feedback 1K+ Reviews
1K+ Reviews


